
Provenence Scientific delivers clinically focused molecular diagnostic solutions that support precision medicine, therapeutic optimization, and advanced pathogen detection. Our approach integrates validated molecular technologies with disciplined laboratory execution to ensure accurate, reproducible, and clinically meaningful results.We work with clinical laboratories, healthcare organizations, and research partners to implement high-complexity testing programs aligned with scientific and regulatory standards.
Pharmacogenomics
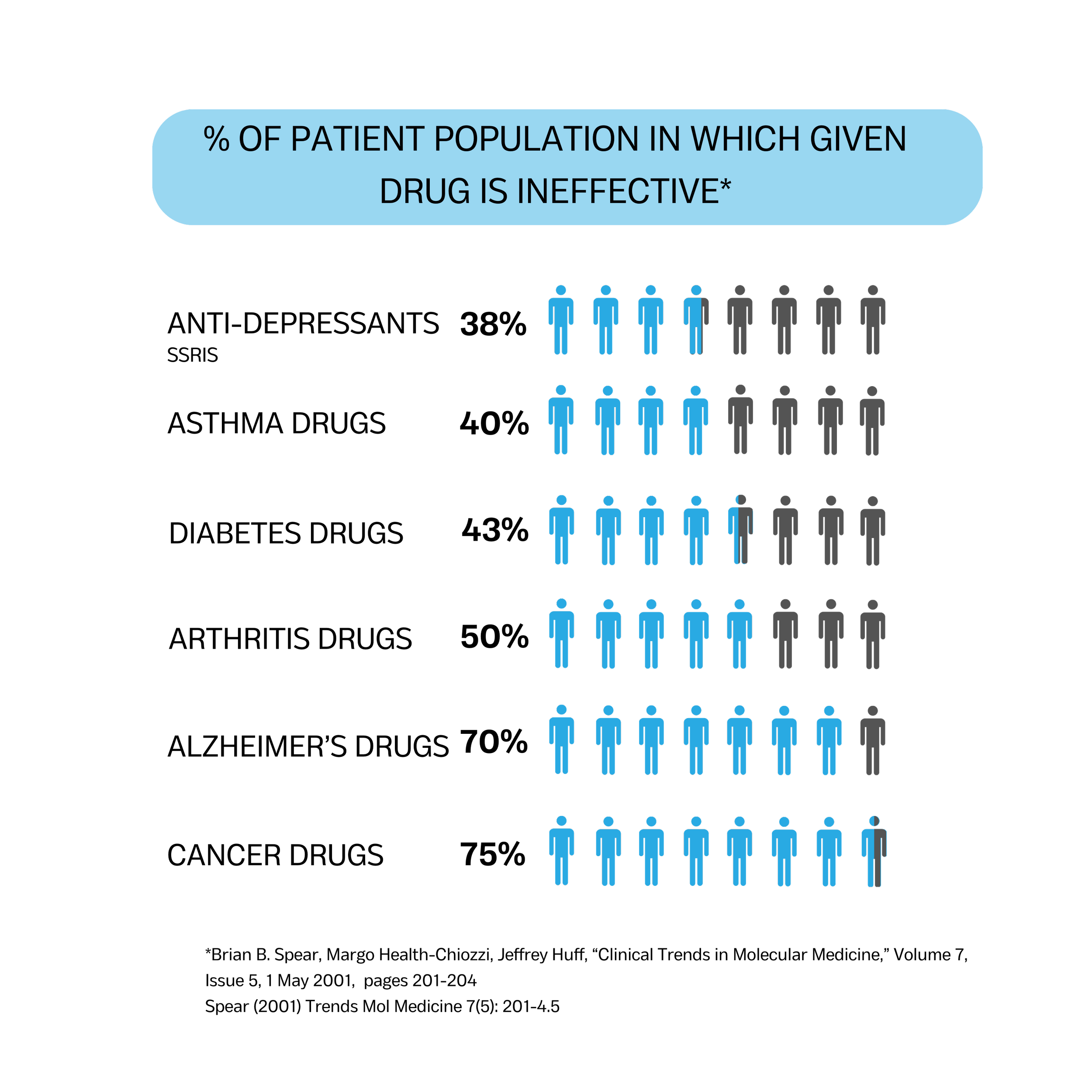

Pharmacogenomic (PGx) testing evaluates inherited genetic variation that influences drug metabolism, therapeutic response, and the risk of adverse drug reactions. Differences in how individuals process medications can significantly affect treatment effectiveness, dosing requirements, and safety profiles. Because PGx results directly inform prescribing decisions, analytical accuracy, sensitivity, specificity, and reproducibility are essential to ensure clinical reliability.Provenence Scientific supports clinical and molecular laboratories in developing and validating robust PGx testing programs. Our approach includes clinically relevant target selection, comprehensive analytical validation design, performance verification, precision and reproducibility assessment, and structured genotype-to-phenotype interpretation frameworks. We also support workflow integration, quality control strategies, and documentation aligned with CLIA high-complexity laboratory standards and regulatory expectations.By emphasizing scientific rigor and operational feasibility, we help laboratories implement pharmacogenomic testing programs that are defensible, compliant, scalable, and ready for clinical deployment.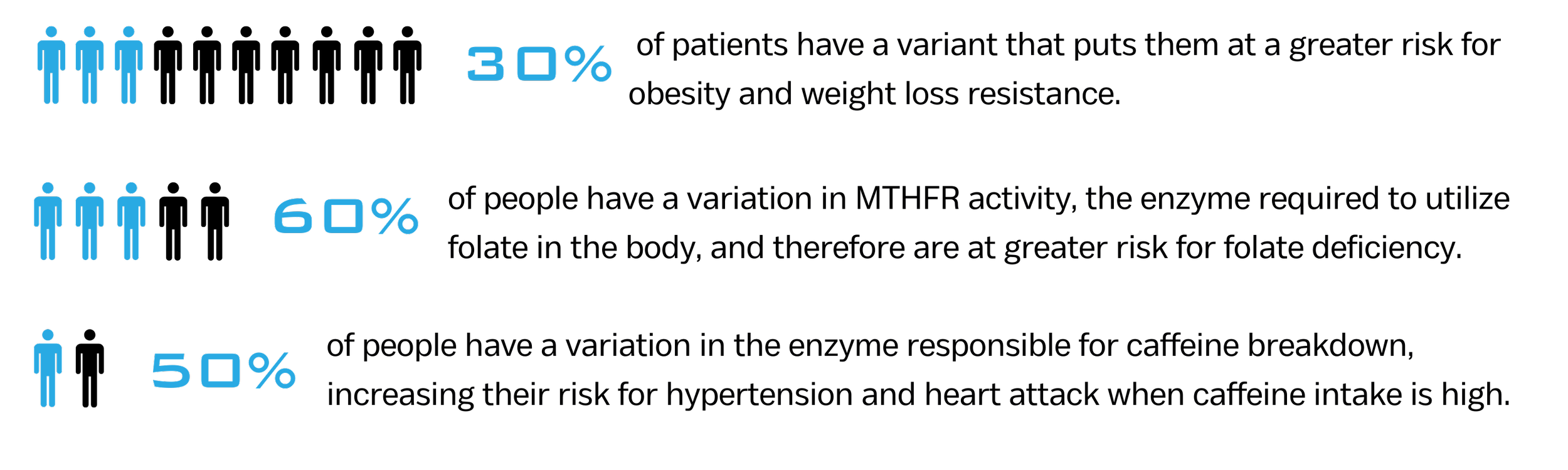
Nutrigenomics
.
Nutrigenomic testing evaluates inherited genetic variation associated with metabolism, nutrient utilization, exercise response, inflammatory pathways, and metabolic regulation. As interest in precision nutrition and lifestyle genomics continues to grow, laboratories require scientifically rigorous and operationally sound validation frameworks to ensure analytical performance and clinical reliability.Provenence Scientific supports clinical and molecular laboratories in the development and validation of nutrigenomic testing programs. Our services include target selection support, analytical validation study design, accuracy and precision assessment, reproducibility testing, performance verification, and workflow optimization aligned with CLIA high-complexity laboratory requirements.We assist laboratories in establishing defensible validation documentation, interpretation frameworks, and quality control processes to ensure testing programs are compliant, scalable, and scientifically credible. Our approach emphasizes analytical integrity, reproducibility, and responsible integration of genomic data into structured reporting models.By combining molecular diagnostics expertise with regulatory awareness and operational experience, we enable laboratories and healthcare institutions to confidently implement nutrigenomic testing services grounded in validated science.Infectious Disease
.

Antibiotic-resistant pathogens and complex polymicrobial infections require diagnostic platforms that are both highly sensitive and operationally robust. Molecular methodologies, including real-time PCR and next-generation sequencing (NGS), enable rapid and precise detection of infectious agents and associated resistance determinants, offering significant performance advantages over conventional approaches.Provenence Scientific supports clinical and molecular laboratories in the development and validation of infectious disease testing programs utilizing molecular technologies. Our services include analytical validation study design, sensitivity and specificity assessment, limit of detection (LoD) determination, reproducibility evaluation, multiplex performance verification, and workflow optimization aligned with CLIA high-complexity laboratory standards.Traditional culture-based diagnostics remain limited by prolonged incubation times, reduced sensitivity for fastidious or slow-growing organisms, and constrained detection in polymicrobial infections. Molecular platforms address these limitations by enabling comprehensive pathogen detection with faster turnaround times and improved analytical performance.We assist laboratories in establishing scientifically defensible validation documentation, quality control strategies, and regulatory-aligned implementation frameworks to ensure infectious disease testing programs are compliant, scalable, and clinically reliable. By combining molecular expertise with operational experience, Provenence Scientific enables laboratories to confidently implement advanced microbial detection technologies that support targeted therapy and responsible antimicrobial stewardship.
